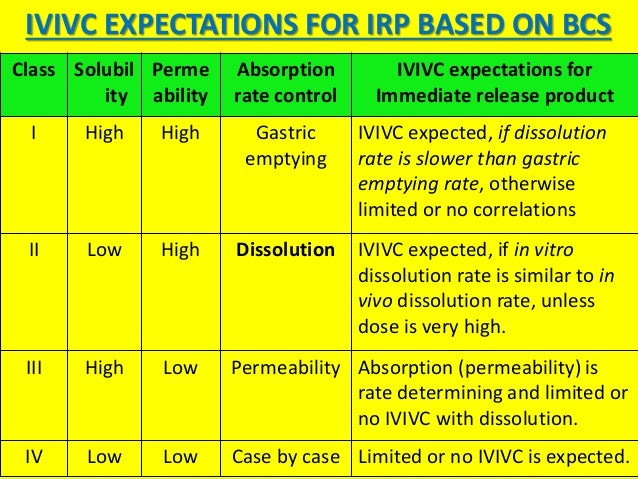
Bcs Classification System
The position of rifampicin with respect to the biopharmaceutics classification system (BCS) was explored on the basis of its aqueous solubility and intestinal permeability. The aqueous solubility was determined between pH 1 and 7 by the conventional shake-flask method. Permeability coefficients of rifampicin and the US FDA listed low (furosemide, ranitidine) and high permeable (caffeine, naproxen) model drugs were determined in the three segments of the rat intestine by employing the everted gut sac model. The samples were analyzed by HPLC. The maximum human single dose of rifampicin (600 mg) was insoluble in 250 ml of aqueous media between pH 3 and 7. The determined apparent permeability coefficient (Papp) values for rifampicin were 4.856×10−6, 2.117×10−6, and 2.149×10−6 cm/sec in the rat duodenum, jejunum and ileum, respectively. These values were similar to those of the low permeable drugs, ranitidine and furosemide, for which Papp values were determined to be 1.767×10−6−2.426×10−6 and 2.469×10−6−3.008×10−6 cm/sec, respectively. The determined values for high permeable model drugs, viz., caffeine and naproxen, were ∼10–20 folds higher than rifampicin or even ranitidine and furosemide. The study suggests that rifampicin is a drug with low solubility and even low intestinal permeability and hence qualifies to be classified in BCS Class IV, instead of Class II where it is being categorized presently. The contention is supported by the reported data on the permeability of the drug through cell monolayers, the mass balance and the absolute bioavailability values in the literature, and the data for rifampicin according to “Lipinski's rule of five”.
Bcs Classification Of Drugs List
Biopharmaceutical Classification System (bcs) Class Ii

The Biopharmaceutics Classification System is a system to differentiate the drugs on the basis of their solubility and permeability. This system restricts the prediction using the parameters solubility and intestinal permeability. The solubility classification is based on a United States Pharmacopoeia (USP) aperture.
Thus it provides security to your data. Free pocket pc software download hk.